And none of them are pretty.
Fluoride is either a fluorine ion, or a compound containing fluorine.



There are four types of fluorine compounds.
- Sodium fluoride (NaF)




What’s the difference between stannous fluoride vs sodium fluoride?
This seems good, but since salivea repairs teeth, it actually sabotages tooth repair.
Fluoride has been shown to induce osteoclastogenesis in mice.
Osteoclasts are cells that degrade bone.
So amine fluoride not only dissolves teeth, it keeps slaivea from repairing teeth!
Olaflor is an amine fluoride,
AmF compounds result in a marked increase in enamel micro hardness when compared to NaF.
7 Side Effects of Consuming Fluoride You Should Know About
- Tooth Discoloration. Consumption of too much of fluoride leads to yellowed or browned teeth. …
- Tooth Decay. High intake fluoridated water can lead to the weakening of enamel. …
- Skeletal Weakness. …
- Neurological Problems. …
- High Blood Pressure. …
- Acne. …
- Seizures.

6 Researched Dangers of Fluoride
1. May cause damage to the brain and central nervous system
One major cause of concern when ingesting fluoride is the potential it may have to negatively affect the central nervous system (CNS). A famed study by Phyllis Mullenix was one of the first occasions on which this CNS effect was quantified. In fact, you couldn’t discover any arguments on the pros and cons of fluoridation that do not include Mullenix’ discovery.
This well-designed study, using the latest technology available at the time, was conducted in the mid-1990s on rats. Emphasis on the study came from reports from China that high levels of fluoride in drinking water (multiples of any current levels in the U.S.) had been seen to affect the CNS preceding skeletal fluorosis. The animals were given varying levels of fluoride at multiple stages of development and compared with controls.
Mullenix discovered that fluoride treatment during fetal, weanling and adult development all had pronounced behavioral effects, even when the plasma (blood) levels of fluoride did not seem to be all that elevated. Prenatal exposures seemed to result in hyperactivity symptoms, whereas weanling/adult exposures both resulted in “cognitive deficits.” (56)
Another brain-related danger of fluoride is the potential that it may result in lower IQ’s. Studies have found varying degrees of IQ disparity, from a 2.5-point drop to a 7-point difference in children exposed to fluoridated water versus those who are not. (57)
The meta-analysis that reflected a 7-point change in IQ was conducted by researchers at the Harvard School of Public Health. Although the results were highly suggestive, the scientists were quick to state that the studies they examined were not conclusive enough to draw any sort of cause-and-effect relationship and, in some cases, reflected fluoride levels in water far above what anyone in the U.S. is generally exposed to. (59)
However, they were intrigued enough by their results to begin a pilot study in China to garner more information. This is the first of what is said to be many follow-up studies regarding fluoride and intelligence. In the 51 human study participants, it was found that moderate-to-severe dental fluorosis was correlated with poorer scores on two types of intelligence tests. (60)
Some are also concerned that the combination of aluminum and fluoride exposure might be a contributing factor to the development of Alzheimer’s disease. (61) In animal studies, rats given sodium fluoride (NaF) had significantly higher tissue aluminum levels and “alterations of cerebrovascular and neuronal integrity.” (62) As there is a large amount of evidence to suggest that aluminum plays a role in the development of Alzheimer’s, it’s an avenue worth looking into deeper. (63, 64)
Since fluoride crosses the blood-brain barrier, there are a number of effects it could have that are currently unknown. However, we do know, according to studies listed above, that fluoride might disrupt circadian rhythms and does alter the levels of certain neurotransmitters that are vital to healthy brain chemistry.
2. Has been associated with moderate cancer risk
Another hotly debated potential risk of fluoride is its potential to influence the risk of certain cancers. (65)
A 1977 study comparing the 10 largest fluoridated and unfluoridated cities at that time found an increase in cancer-related deaths of 18 percent in the fluoridated cities compared to those that weren’t, equating to about 3,000 more cancer deaths per 10 million persons in 1969, the studied year. (66)
A similar study, published earlier in 1977, detailed a review of cancer death rates over 17 years, between 1952–1969. No difference in the rates was discovered in persons up to 44 years of age between fluoridated and unfluoridated communities. In those between 45–64 years of age, an additional 1,500 cancer deaths per 10 million people was recorded in fluoridated cities, and the number rose to 3,500 more cancer deaths per 10 million people when observing those over 65. (67)
One review of cancer instances between 1978–1992 found that:
Cancers of the oral cavity and pharynx, colon and rectum, hepato-biliary and urinary organs were positively associated with FD [fluoridated drinking water]. This was also the case for bone cancers in male, in line with results of rat experiments. Brain tumors and T-cell system Hodgkin’s disease, Non-Hodgkin lymphoma, multiple myeloma, melanoma of the skin and monocytic leukaemia were also correlated with FD.
In that study, four types of cancers were actually correlated with a decreased risk in persons exposed to fluoridated water. (68)
Of particular interest in the fluoride debate is the instances of osteosarcoma, a rare form of bone cancer. A relatively small study was published in 1993 finding a 6.9 times increased risk of osteosarcoma among males under 20 years of age in the most fluoridated parts of a 3-county area. (69)
The Harvard School of Dental Medicine also conducted a study about this risk and found similar results of an increased risk for young males of osteosarcoma when drinking fluoridated water. (70)
However, other follow-up studies have found no link or at least no significant increase in osteosarcoma cases between fluoridated and unfluoridated communities. (71, 72, 73)
3. Could increase risk of bone fractures
Unlike what was originally assumed, it seems the ingestion of fluoride does not have a positive impact on bone health, and may actually have a pronounced negative effect.
A study conducted in Mexico found an increase in bone fractures and major tooth damage in children exposed to fluoridated water. (74)
Other studies have found an increase in hip fracture risk among the elderly when consistently drinking water fluoridated at 1 ppm. (75) Other sources find no link between fluoridation and bone fractures. (76)
It’s true that the rate of hip fractures related to osteoporosis has increased in the elderly over the same period of time as the dawn of water fluoridation, but there is not yet evidence to suggest this could be attributed in any way to fluoride, as the causative factors behind such a disease are vast and not usually just one toxin or risk factor. (77)
4. Associated with hypothyroidism
Possibly related to the association between fluoride and hormonal function is the evidence that fluoride may be linked to hypothyroidism. In a study comparing fluoridated and unfluoridated areas of the U.K., researchers discovered that those living in unfluoridated areas were almost two times less likely to develop hypothyroidism. (78)
5. May interfere with sexual development
Remember that fluoride has been found to collect in the pineal gland? This may have further-reaching effects than just the interference with circadian rhythms. A 1997 study using gerbils discovered that fluoride was associated with faster sexual development in the females in the study. (79)
While these results haven’t been tested further in humans, it could be a very important point, as early puberty may possibly lead to issues ranging from short stature to an increased risk of breast cancer.
6. Associated with diabetes risks
As diabetes diagnoses are at an all time high, a great deal of research is focused on the ways we can reduce the impact of this reversible condition. (80) A literature review of the connection between fluoride and diabetes, conducted by Dr. Geoff Pain, an Australian chemistry specialist, left the scientist in no doubt of the results. Pain stated, (81)
There is strong evidence that fluoride causes diabetes … Diabetics are a “sensitive subpopulation” or “vulnerable group” and no attempt has been made by Australian health authorities to warn diabetics about fluoride toxicity or protect them from harmful exposure.
However, this review does not equate to convincing proof that diabetes truly is caused by fluoride exposure. While it warrants further investigation, another study found that low levels of fluoride, like those found in water in the U.S., actually helps to improve insulin resistance and aids in glucose homeostasis. (82)
There hasn’t been a single US study of fluoridation, prenatal exposure and natal development,”
Side effects
Calcium Fluoride versus Industrial Fluoride
There is a big difference between naturally occurring Calcium fluoride and Industrial fluoride. Natural calcium fluoride is considered insoluble (to 8–10 ppm fluoride maximum depending on water pH). But industrial synthetic fluorides are fully soluble and are all toxic calcium chelators.
J Environ Public Health. 2013; 2013: 439490.Published online 2013 Jun 6. doi: 10.1155/2013/439490PMCID: PMC3690253PMID: 23840230
Physiologic Conditions Affect Toxicity of Ingested Industrial Fluoride
Synthetic industrial fluoride compounds lack calcium and are listed toxic substances
Calcium fluoride CaF2 is found in natural minerals and is not labeled a toxic compound because of the comparatively high lethal oral acute dose of the purified compound when tested in mammals (LD50 ~ 3,750 mg/kg).
The fluoride compounds, sodium fluoride NaF and fluorosilicic acid H2SiF6, added into municipal water for human ingestion purposes are synthesized artificially by industrial reaction and have been used as rodenticides, insecticides, and pediculicides, with acute oral lethal doses in experimental animals comparable to arsenic and lead (LD50 ~ 125 mg/kg) (The Merck Index [4]) due to the fluoride at ~60–90 mg/kg.
Fluoride is not a normal constituent of the mammalian bloodstream (Merck manual for Health Care Professionals [6]). It has no nutritive value [7] or physiologic function but has been believed by some to be useful for teeth based on an initial correlation with natural calcium fluoride in drinking water [1, 8]. The chief ingredient in normal teeth enamel is hydroxyapatite that contains calcium phosphate, not fluoride.
The reported adverse consequences of adding fluoride lacking calcium into public water supplies include effects on man, animals, and the environment [1, 8–12]. Ingested industrial fluoride incorporates chiefly into bone with an ion exchange process that is irreversible and thus not physiologic. Normal biochemical effects of nutrient minerals are saturable and readily reversible. Fluorine leads all elements in electronegativity and is extremely reactive and not found in nature. But fluoride is permanent because the ion has no electronegativity, cannot be reduced further, or oxidized by any known substance. Fluoride instead associates with positive charged ions in particular aluminum, calcium, and iron. Thus its toxicity depends on the environment in which it resides.
Soluble fluoride at 60 mg/kg single oral dose without calcium causes acute heart failure in research animals (CDC [5]) and caused lethal heart failure reported in a child after swallowing concentrated dental gel [13]. Twenty-five ppm artificial fluoridated water leads to chronic heart failure in research animals [5] which compares with levels during accidental overfeeds where kidney dialysis patients died (Gessner et al. [14]). At lower concentrations (~1 ppm), artificially fluoridated water supplies are documented to have caused horses, frogs, chinchillas, and alligators to die prematurely that consumed treated water continuously for extended periods of time (Spittle [12]). Discharged fluoride into the Columbia River to ~0.3 ppm blocked salmon navigation upstream to spawn (Damkaer and Dey [15]). Even though natural fluoride at 1 ppm is in the world’s oceans with substantial calcium and magnesium salts, this arrangement is normal and harmless to aquatic species.
Natural calcium fluoride is considered insoluble (to 8–10 ppm fluoride maximum depending on water pH). But industrial synthetic fluorides are fully soluble and are all toxic calcium chelators. The degree of absorption of any fluoride compound after ingestion is correlated with its solubility (Goodman and Gilman [16]). Industrial fluorides are completely absorbed, but natural fluoride minerals cryolite (Na3AlF6) or fluorite (mineral fluorspar with CaF2) are poorly absorbed (see Endnote 1). The dietary cations calcium and iron retard absorption by forming complexes in the GI tract. Although large populations are reported to safely consume 1 ppm fluoride in water for long periods of time, this is when it exists naturally at this level.
In what was considered unthinkable, in Hooper Bay, Alaska, in 1994 an industrial fluoridation overfeed of calcium-deficient Yukon River drinking water caused fatal heart block in an otherwise healthy 41 year old male. This is the largest known poisoning in the U.S. from a fluoridated water supply. Approximately 300 people with severe gastrointestinal pain survived the incident (Gessner et al. [14]). Electronic feeding equipment is now employed to prevent overfeeds and acute poisoning. But chronic effects of industrial fluoridation of public water supplies on humans, animals, and the environment require further study if fluoridation of fresh water supplies continues. Many countries require great expenditures to remove endogenous natural fluoride from drinking water that causes skeletal and other pathology at 8–10 ppm even when water contains substantial antidote calcium to minimize assimilation of the ingested fluoride [5]. The present study investigates conditions involved in acute and chronic fluoride toxicity and environmental effects of industrial fluorides added into public water.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3690253/
Calcium fluoride is the inorganic compound of the elements calcium and fluorine with the formula CaF2. It is a white insoluble solid. It occurs as the mineral fluorite (also called fluorspar), which is often deeply coloured owing to impurities.

The mineral fluorite is abundant, widespread, and mainly of interest as a precursor to HF. Thus, little motivation exists for the industrial production of CaF2. High purity CaF2 is produced by treating calcium carbonate with hydrofluoric acid:[10]CaCO3 + 2 HF → CaF2 + CO2 + H2O
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
Applications
Main article: Fluorite
Naturally occurring CaF2 is the principal source of hydrogen fluoride, a commodity chemical used to produce a wide range of materials. Calcium fluoride in the fluorite state is of significant commercial importance as a fluoride source.[11] Hydrogen fluoride is liberated from the mineral by the action of concentrated sulfuric acid:[12]CaF2 + H2SO4 → CaSO4(solid) + 2 HF
Niche uses
Calcium fluoride is used to manufacture optical components such as windows and lenses, used in thermal imaging systems, spectroscopy, telescopes, and excimer lasers. It is transparent over a broad range from ultraviolet (UV) to infrared (IR) frequencies. Its low refractive index reduces the need for anti-reflection coatings. Its insolubility in water is convenient as well. Doped calcium fluoride, like natural fluorite, exhibits thermoluminescence and is used in thermoluminescent dosimeters. It forms when fluorine combines with calcium.
Safety
CaF2 is classified as “not dangerous”, although reacting it with sulfuric acid produces very toxic hydrofluoric acid. With regards to inhalation, the NIOSH-recommended concentration of fluorine-containing dusts is 2.5 mg/m3 in air.[10]
https://en.wikipedia.org/wiki/Calcium_fluoride
Fluorine is a chemical element with the symbol F and atomic number 9. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium.
| Table 1. Plants sensitive to fluoride.* | ||
| Common Name | Scientific Name | Notes |
| Apricot | Prunus armeniaca | Some cultivars are intermediate insensitivity. |
| Box Elder | Acer negundo | |
| Blueberry | Vaccinium corymbosum | |
| Sweet corn | Zea mays | |
| Douglas-fir | Pseudotsuga menziesii | |
| Gladiolus | Gladiolus sp. | Bract and leaf injury. |
| Grape | Vitis vinifera | V. labrusca is intermediate. |
| Oregon Grape | Mahonia aquifolium | |
| Western Larch | Larix occidentalis | |
| Peach | Prunus persica | Soft suture or red suture disease of the fruit. |
| Pine | Pinus sp. | Eastern white pine, lodgepole, scotch, Mugo, ponderosa |
| Plum | Prunus domestica | Flowering plums are resistant. |
| Blue Spruce | Pice pungens | |
| Tulip | Tulipa sp. | |
| Corn plant | Dracaena sp. | Most species and cultivars. |
| Yucca | Yucca sp. | |
| Spider plant | Chlorophytum comosum | |
| Tahitian Bridal Veil | Gibasis pellucida | |
| Lily | Lilium spp. | |
| * Plant are considered sensitive when injury has been observed on most of the species and when leaf analysis revealed a fluoride content less than 50 ppm. |
Olaflor

Olaflur (INN, or amine fluoride 297) is a fluoride-containing substance that is an ingredient of toothpastes and solutions for the prevention of dental caries.[1] It has been in use since 1966. Especially in combination with dectaflur, it is also used in the form of gels for the treatment of early stages of caries, sensitive teeth, and by dentists for the refluoridation of damaged tooth enamel.[2]
Overdosage leads to irritation of the oral mucosa. In especially sensitive persons, even standard doses of olaflur can cause irritation.[2] Like other fluoride salts, olaflur is toxic when given in high doses over an extended period of time. Especially in children, before the development of the permanent teeth, overdosage can lead to dental fluorosis, a discolouring and weakening of the enamel.[3] In acute cases of overdosage, for example when an olaflur containing preparation is swallowed, calcium in any oral form serves as an antidote. Often milk is used because it is usually at hand.[2]
Interactions
Because calcium fluoride is practically insoluble in water, calcium-containing drugs and food inhibit the action of olaflur.[2]
Chemistry and mechanism of action
Olaflur is a salt consisting of an alkyl ammonium cation and fluoride as the counterion. With a long lipophilic hydrocarbon chain, the cation has surfactant properties. It forms a film layer on the surface of teeth, which facilitates incorporation of fluoride into the enamel. The top layers of the enamel’s primary mineral, hydroxylapatite, are converted into the more robust fluorapatite. The fluoridation reaches only a depth of a few nanometres, which has raised doubts whether the mechanism really relies on the formation of fluorapatite.[4]
Synthesis[edit]
The synthesis of olaflur starts from cattle’s tallow.[5] The contained fatty acids, mainly stearic acid (C17H35COOH), are obtained by hydrolysis, and then converted to the corresponding amides, which in turn are reduced catalytically to the primary amines (largely octadecylamine). Addition of acrylonitrile, followed by another reduction, yields N-alkyl-1,3-propanediamines. The two nitrogen atoms react with ethylene oxide to form tertiary amines. Finally, hydrofluoric acid is added to give the end product. Because olaflur is produced from a mixture of fatty acids, some molecules have side chains that are longer or shorter than 18 carbon atoms. Other byproducts of the reaction include hydroxyethyl ethers resulting from addition of ethylene oxide to the free hydroxyl groups. The presence of these side products is clinically irrelevant.[5]
https://en.wikipedia.org/wiki/Olaflur
Dokter Hans Moolenburgh sr. over de illegale fluoridering in Curaçao
Gepubliceerd op 23 mei 2016
Rudolf de Wit interviewt Dokter Moolenburgh over de illegale fluoridering van het drinkwater in Curaçao, sinds 1976 ongrondwettelijk, en de ernstige gevolgen die dit enzymgif voor de volksgezondheid kan hebben, terwijl het beweerde nut (voor een kleine doelgroep van kinderen) nooit afdoend bewezen is. De dwangmedicatie is een overtreding van de Mensenrechten. Lees meer op https://www.dodelijkeleugens.nl/schending-mensenrechten-massamedicatie/
- Categorie
- Licentie
- Standaard YouTube-licentie
Schending mensenrechten door illegale massamedicatie
Mensenrechten geschonden door onwettige massamedicatie met fluoride
door C.F. van der Horst
Sinds 1962 wordt het drinkwater in Curaçao gefluorideerd; de maatregel werd in 2006 in de Landsverordening Drinkwater officieel vastgelegd, terwijl de aanleiding voor Artikel 11 van de Nederlandse Grondwet (van kracht sinds 1983) en het daarop gebaseerde Artikel 13 van de Staatsregeling van Curaçao juist het voorkomen van massamedicatie zoals fluoridering was. De fluoridering is daarom illegaal en zoals u hieronder zult lezen, een overtreding van de Mensenrechten.
Is het niet vreemd dat Nederland Mensenrechten hoog in het vaandel heeft, maar deze schending door massamedicatie in het eigen Koninkrijk ongemoeid laat, terwijl sinds 1983 fluoridering zelfs een overtreding van de Grondwet is?
Neurotoxine
De veronderstelde noodzaak voor de dwang- en massamedicatie is dat fluoride het tandglazuur zou verharden waardoor cariës (tandbederf) bij kinderen wordt teruggedrongen. Dit nut staat echter niet vast. Zo stelde het CDC (Centers for Disease Control and Prevention, het Amerikaanse agentschap belast met het opsporen en onderzoeken van de volksgezondheid trends): “Het aantreffen van tandbederf in een populatie is niet omgekeerd evenredig met de concentratie van fluoride in het tandglazuur en een hogere concentratie aan fluoride in het tandglazuur is niet noodzakelijkerwijs doeltreffend in het voorkomen van tandcariës.” Tegenstanders beweren dat als er al voordelen zouden zijn, deze niet opwegen tegen de bewezen giftigheid. Het gerenommeerde Britse artsenblad The Lancet berichtte in 2014 dat fluoride als een neurotoxine wordt beschouwd en dat de stof als zodanig bij kinderen neurologische ontwikkelingsstoornissen zoals autisme, hyperactiviteit, dyslexie en andere cognitieve stoornissen kan veroorzaken, zoals het uit zo’n 50 andere onafhankelijke studies gebleken significant dalen van het IQ bij kinderen die aan fluoride blootgesteld zijn.
Los van de discussie over veiligheid en effectiviteit schendt de gebruikersdwang bij de preventieve gezondheidsmaatregel die fluoridering beweerd wordt te zijn, essentiële wetten en rechten.
Geen voedings- maar geneesmiddel
Fluoride is een lichaamsvreemde stof. Het maakt geen deel uit van enig stofwisselingsproces en het is daarom geen voedingsmiddel. Dit is de conclusie van zowel de Europese voedselwaakhond EFSA als de Amerikaanse FDA. Tandbederf ontstaat NIET als gevolg van een gebrek aan fluoride. Fluoridering van het drinkwater is derhalve geen aanvulling van een tekort in het dieet, zoals soms vitaminen en mineralen die aan voedingsmiddelen worden toegevoegd. Integendeel, fluoride (met inbegrip van de fluoridezouten die aan water worden toegevoegd, hexafluorkiezelzuur en natriumsilicofluoride) wordt geclassificeerd als een medicijn. Deze vaststelling maakt de toevoeging van fluoride aan drinkwater tot massamedicatie.

Schending van de Grondwet en Staatsregeling
Artikel 11 van het Statuut voor het Koninkrijk der Nederlanden luidt:
“1. Voorstellen tot verandering in de Grondwet, houdende bepalingen betreffende aangelegenheden van het Koninkrijk, raken Aruba, Curaçao en Sint Maarten.”
De aanpassing van de Nederlandse Grondwet op 19 januari 1983 is tot op heden echter in Curaçao genegeerd. Het gaat om Artikel 11, getiteld Grondrecht, onaantastbaarheid menselijk lichaam:
“Ieder heeft, behoudens bij of krachtens de wet te stellen beperkingen, recht op onaantastbaarheid van zijn lichaam.”
Ofschoon de Staatsregeling van Curaçao (de nationale grondwet van het eiland) Artikel 11 van de Nederlandse Grondwet in zijn geheel en ongewijzigd overgenomen heeft in haar Artikel 13, gaat de Landsverordening Drinkwater hier volledig aan voorbij. De Memorie van Toelichting behorende bij de Staatsregeling meldt nota bene:
“Bij het recht op onaantastbaarheid van het lichaam gaat het om het recht op afweer van invloeden van buitenaf op het lichaam. Het bevat twee terreinen: het recht te worden gevrijwaard van schendingen van en inbreuken op het lichaam door anderen en het recht zelf over het lichaam te beschikken.”
De massamedicatie door fluoridering schendt de onaantastbaarheid van het lichaam al 33 jaar. Derhalve is de Landsverordening Drinkwater van 2006 (een wet ondertekend door Minister Smith van Volksgezondheid en Sociale Ontwikkeling, Minister Dick van Justitie en Minister Leeflang van Algemene Zaken en Buitenlandse betrekkingen) die de gebruikersdwang van fluoride oplegt in strijd met zowel de Grondwet als de Staatsregeling!

Jurisprudentie
Er bestaat jurisprudentie inzake massamedicatie en fluoridering in het Koninkrijk der Nederlanden. Artikel 11 in de Grondwet hervorming van 1983 was een direct gevolg van de fluorideringskwestie die in de 60er en 70er jaren in Nederland speelde. Auteur Dr. Dennis Edeler schreef hierover: “In vervolg op het Fluorideringsarrest van de Hoge Raad [op 22 juni 1973] is in 1983 met de algehele wijziging van de Grondwet ook het sociale grondrecht opgenomen van de persoonlijke integriteit. Op grond van de jurisprudentie tast fluoridering het menselijk lichaam aan en is vanuit dit perspectief strijdig met de onaantastbaarheid.”
Desgevraagd meldt dokter Hans Moolenburgh sr.: “Artikel 11 is opgesteld door professor Auke Bloembergen, voormalig hoogleraar Burgerlijk Recht aan de Rijksuniversiteit Leiden en lid van de Hoge Raad, en het VVD kamerlid mr. Annelien Kappeyne van de Coppello op initiatief van mr. Hector van Fenema (oud-burgemeester van Zandvoort), mr. Foppe Oberman, advocaat, en mijzelf, destijds hoofd van de landelijke anti-fluorideringsbeweging.” Hiermee is bewezen dat de fluoridering aanleiding is geweest tot dit Grondwet Artikel.
Mensenrechten met voeten getreden
Niet alleen de Grondwet en Staatsregeling maar ook de Mensenrechten worden door de massamedicatie met fluoride met voeten getreden. De Universele Verklaring van de Rechten van de Mens zegt o.a.:
Artikel 3
Een ieder heeft het recht op leven, vrijheid en onschendbaarheid van zijn persoon.
Artikel 19
Een ieder heeft recht op vrijheid van mening en meningsuiting. Dit recht omvat de vrijheid om zonder inmenging een mening te koesteren en om door alle middelen en ongeacht grenzen inlichtingen en denkbeelden op te sporen, te ontvangen en door te geven.
Dwangmedicatie met fluoride is een schending van iemands persoon en gaat volledig aan zijn of haar mening voorbij.
Art. 43 van het Statuut Koninkrijk, waarborg mensenrechten Nederland/Ned. Antillen/Aruba luidt:
“1. Elk der landen draagt zorg voor de verwezenlijking van de fundamentele menselijke rechten en vrijheden, de rechtszekerheid en de deugdelijkheid van het bestuur.”
Vanuit regeringswege zou men derhalve op de mensenrechten moeten toezien. Dat fluoridering in 2006 in een Landsverordening vastgelegd is, laat onverlet dat het om een schending van de Mensenrechten gaat.
Schending van de Code van Neurenberg
De Code van Neurenberg werd in 1947 in het leven geroepen om de vreselijke medische experimenten die Nazi-artsen hadden uitgevoerd, te voorkomen. Het zonder medisch toezicht en zonder controle op de individuele dosis jarenlang toedienen maakt de massamedicatie door fluoridering tot een slecht opgezet en uitgevoerd medisch experiment. Artikel 1 van de Code van Neurenberg zegt over medische experimenten: “De vrijwillige toestemming van de proefpersoon is absoluut noodzakelijk.” En voorts dat het individu “zonder tussenkomst van enig element van geweld, fraude, bedrog, pressie, misleiding of enige andere verborgen vorm van beperking of dwang” kan worden gevraagd om aan het experiment deel te nemen. Door iedereen te verplichten aan het medisch experiment van gefluorideerd water mee te doen (gebruikersdwang) wordt de Code op grove wijze overtreden.

Voorzorgsbeginsel
Wat betreft gezondheidsrisico’s zou het voorzorgsbeginsel te allen tijde van toepassing moeten zijn. Populair gezegd komt het neer op voorkomen is beter dan genezen. In opdracht van de Gezondheidsraad, het hoogste adviesorgaan voor de Nederlandse regering, is in 2003 een studie naar het voorzorgsprincipe uitgevoerd. Daarin zegt men o.a.: “In het algemeen wordt het voorzorgbeginsel toepasbaar geacht als er potentiële risico’s zijn dat non-negligible damage wordt veroorzaakt.” Dat is zeker het geval bij fluoride waar verschillende onafhankelijke wetenschappers hebben gewaarschuwd tegen gevaren voor de gezondheid. Deze variëren van ziekteverschijnselen die al kort na de fluoridering optreden, zoals zweertjes in de mond, huidafwijkingen, hoofdpijnen, visuele storingen, etc., tot de veel gevaarlijkere op de lange termijn optredende, zoals botafwijkingen, aandoeningen van het centrale zenuwstelsel, ADHD, autisme, diabetes, migraine, hartfalen en kanker. De bezorgdheid over deze risico’s is nog nooit afdoend weggenomen.
Daarbij is het onmogelijk een standaard dosis voor ieder afzonderlijk persoon te bepalen en stelt men door de gebruikersdwang van fluoride zelfs kwetsbare leeftijdsgroepen (zuigelingen, peuters en ouderen) aan de massamedicatie bloot.
Uit voorzorg moet men alle lichaamsvreemde stoffen, en om de bovengenoemde associaties zeker fluoride, uit water en voeding weren.
Volwaardige voeding: een risicoloos alternatief
De voornaamste doelgroep van fluoridering zijn kinderen, met name in de lagere inkomensklassen. Dat iedereen wordt gedwongen fluoride in te nemen terwijl slechts een klein gedeelte van de bevolking baat erbij zou hebben, is bevreemdend.
Bovendien is bewezen dat cariës uitsluitend wordt veroorzaakt door het geraffineerde voedingspakket (men leze het werk van tandarts Weston Price). Een causale (oorzakelijke) benadering van een kwestie is natuurlijk veel beter dan een symptomatische. Derhalve zou — in plaats van dwangmedicatie voor iedereen — gerichte aandacht aan de juiste doelgroepen voor juiste voeding een veel logischer en veiliger oplossing zijn.
Mensen die desalniettemin voor fluoride kiezen kunnen hiervoor terecht bij hun tandarts of een gefluorideerde mondspoeling aanschaffen.
Het boek Dodelijke leugens. Artsen en patiënten misleid beschrijft de ontdekkingen van een aantal wetenschappers over de schadelijkheid van fluoride en wat men zelf kan ondernemen om kinderen te helpen sterke tanden te krijgen en houden.
Tot slot: dokter H.C. Moolenburgh sr.
Dokter Hans Moolenburgh voerde met zijn artsengroep in de zeventiger jaren dubbelblinde proeven uit naar de effecten van gefluorideerd drinkwater op de menselijke gezondheid. De dokters vonden diverse klachten — zoals heel nare buikpijn (vooral de hele nacht gillende baby’s waren opvallend), hoofdpijn en astmatische verschijnselen — die alleen en blijvend verdwenen door het drinken van ongefluorideerd water. De tomeloze inzet van de medicus en zijn kleine groep toegewijde medewerkers voor de Rechten van de Mens heeft ertoe geleid dat de dwangmedicatie in Nederland in 1976 werd stopgezet. Om gebruikersdwang door de toevoeging van fluoride of enige andere medicatie aan het drinkwater te voorkomen, heeft hij daarenboven — zoals eerder vermeld — zich sterk gemaakt voor de uitbreiding van de Grondwet.
Dokter Moolenburgh heeft dit artikel gelezen en zijn volledige goedkeuring eraan gehecht. In de video hieronder wordt de fluoride expert geïnterviewd door Rudolf de Wit, de oprichter van de Komishón Anti-Fluoride op Curaçao.
Meer informatie vindt u op FLUORIDE: ZEGEN OF VLOEK? en in het boek Dodelijke leugens. Artsen en patiënten misleid.
Hoe kan een illegale maatregel als fluoride in het drinkwater zo maar worden ingevoerd? Wat is het zogenaamde “draaideur” fenomeen? Wat kunt u doen om u te wapenen tegen fluoride en soortgelijke “gezondheidsbevorderende” maatregelen?
Krijg de feiten. Koop en lees nu het boek Dodelijke leugens. Artsen en patiënten misleid.
Wilt u allereerst een kennismaking? Vraag nu gratis het eerste hoofdstuk aan!
Children Tend to Suffer the Greatest Health Effects
Water fluoridation came about as the result of a massively successful PR campaign, originally aimed to protect aluminum and steel producers from lawsuits against the fluorine pollution coming from their plants. Fluoride is in fact a toxic substance that accumulates in the human body over time, where it has been shown to wreak havoc with enzymes and produce a number of serious adverse health effects, including neurological and endocrine dysfunction. No less than 25 human studies have also linked fluoride with lowered IQ in children, including recent research from Harvard. Approximately 50 percent of the fluoride that you ingest each day ends up accumulating in your bones over a lifetime.
Making matters worse, water fluoridation disproportionately harms young children, as they tend to suffer the greatest health effects. One important point to remember, which few pediatricians, dentists or other health professionals stress, is that you should NOT use fluoridated water when mixing infant formula.
According to Dr. David Kennedy, who produced and directed the documentary film Fluoridegate — An American Tragedy:
“One of California’s highest paid and most prolific Fluoridation advocate admits that giving an infant a formula made with fluoridated tap water will overdose the baby and cause the teeth to come in spotted and fluorotic. One can only wonder why such insanity persists in our country when it has been banned in so many other more advanced democracies.”
http://articles.mercola.com/sites/articles/archive/2013/04/11/portland-fluoridation.aspx
Hidden fluoride in tea and other foods and beverages
Can certain tea preparations be hiding potentially toxic levels of fluoride? You may want to warn your patients with heavy tea habits to consider their overall exposure.Cathy Hester Seckman, RDHMay 1st, 2019
© Natthapon Ngamnithiporn – Dreamstime.com
Dental hygienists are pretty knowledgeable about fluoride. We apply fluoride treatments under ADA guidelines during recare appointments. We recommend different levels of at-home fluoride therapy depending on circumstances and need. We know the approved parts per million in community water supplies. Beyond that, we know which communities in our practice areas have fluoridated water supplies, and which do not have that benefit. We’re aware of fluorosis and its causes.
But did you know fluoride can be present in potentially toxic levels in tea? I was leafing through one of my mother’s less-than-reputable women’s magazines when I read that fluoride found in instant tea mixes can be more than 200% of the recommended safe level. According to the magazine, we should drink only green tea, because fluoride levels in green tea are much lower than in black and instant teas.
Being a typical hygienist, I went straight home to look it up. A cursory Internet search turned up PubMed studies, USDA research papers, and newspaper articles on high fluoride levels in tea. I started wondering about the possible links between tea’s high fluoride levels and fluorosis.
Suppose you have a tea-drinking patient who is in her 50s, with a family history of osteoporosis, and lives in a fluoridated community. On a daily basis, she drinks large quantities of iced tea made from mixes at her favorite coffee shop. This article will give you the background you need to discuss her tea consumption.
How does it get there?
How, exactly, does fluoride get into tea? It all starts with tea plants. Camellia sinensis (var. sinensis) and Camellia sinensis (var. assamica)are the varieties usually grown today. All types of tea—white, yellow, green, oolong, dark, black, and pu-erh—come from these two plants. (Remember that herbal teas are not made from tea plants, but from herbs.) The age of the tea leaves and the fermenting processing differ for each kind of tea. Dark, black, and pu-erh tea would typically be made from older leaves.1
Tea plants are known as fluoride hyperaccumulators, which means they absorb potential toxins and heavy metals to a greater concentration than is in the soil surrounding them.2 The older individual tea leaves get, the more fluoride they can absorb. The fluoride is then released during tea infusion. Bioavailability is close to 100%, because the GI tract readily absorbs soluble fluoride.3
Older tea leaves are also used to make less expensive tea.4 An article in ScienceDirect described a study in the United Kingdom of economy supermarket-branded teas. It was determined that drinking these cheaper teas made from older leaves carried a risk of high exposure to fluoride, up to 150% of the dietary reference intake level.5
Other sources of fluoride
Other fluoride hyperaccumulators include fruit juice, crab, fish, chicken, and rice, but the amounts of fluoride in those foods are much less than in tea.6
We already know our fluoride intake comes from naturally occurring and community fluoridated water, plus toothpastes and mouthwashes, plus recommended supplements. The Public Health Service recommends community water fluoridation at optimum levels ranging from 0.7 ppm to 1.2 ppm (1 ppm is equal to 1 mg/L). The U.S. Environmental Protection Agency has set maximum allowable fluoridation at 4 ppm with a secondary limit at 2 ppm. The American Dental Association, American Academy of Pediatric Dentistry, and the American Academy of Pediatrics jointly recommend guidelines that range from none for infants to 1 mg/day for adults, depending on availability of fluoridated water. The Institute of Medicine recommends a tolerable upper intake at 10 mg/day for those over nine years old.7
- Strained applesauce baby food – 0.01
- Blueberry muffin – 0.39
- Light beer – 0.45
- Coffee, brewed – 0.91
- Chamomile herb tea, brewed – 0.13
- Black tea, brewed – 3.73
- Green tea, brewed – 1.15
- Instant tea powder, unsweetened, dry – 897.72
- Instant tea powder, unsweetened, prepared – 3.35
Effects of excessive fluoride
A 2011 study in the Journal of Clinical Endocrinology and Metabolism concluded that skeletal fluorosis “can result from chronic consumption of large volumes of brewed tea” and that “daily consumption of 1-2 gallons of instant tea can lead to skeletal fluorosis.”10
Tea is supposed to be good for us. It has flavonoids, a mild amount of caffeine, and has been shown to reduce risk for cardiovascular disease, high cholesterol, hypertension, and type 2 diabetes. It’s an anti-inflammatory and an antioxidant.11 Now, it appears, it could also be dangerous in certain circumstances. We, as hygienists, are the health-care workers patients trust to keep them informed about fluoride. When you discover a heavy tea habit in a patient, be sure they’re aware of the risks to their overall health.
References
- Wikipedia. Camellia sinensis. https://en.wikipedia.org/wiki/Camellia_sinensis
- Collins English Dictionary. Definition of hyperaccumulator. https://www.collinsdictionary.com/dictionary/english/hyperaccumulator
- Waugh DT, Potter W, Limeback H, Godfrey M. Risk assessment of fluoride intake from tea in the Republic of Ireland and its implications for public health and water fluoridation. Int J Environ Res Public Health. 2016 Mar; 13(3): 259.
- NHS. Do fluoride levels in cheap tea pose a health risk? July 25, 2013. https://www.nhs.uk/news/food-and-diet/do-fluoride-levels-in-cheap-tea-pose-a-health-risk/
- Chan L, Mehra A, Saikat S, Lynch P. Human exposure assessment of fluoride from tea (Camellia sinensis L.): A UK based issue? Food Res Int. 2013 May;51(2):564-570. https://www.sciencedirect.com/science/article/pii/S0963996913000446
- Oregon State University. Linus Pauling Institute. Fluoride. https://lpi.oregonstate.edu/mic/minerals/fluoride#food-beverage-sources
- Centers for Disease Control and Prevention. Recommendations for using fluoride to prevent and control dental caries in the United States. https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5014a1.htm#tab1
- US Department of Agriculture. USDA National Fluoride Database of Selected Beverages and Foods, Release 2. https://www.ars.usda.gov/ARSUserFiles/80400525/Data/Fluoride/F02.pdf
- ScienceDirect. Skeletal fluorosis. https://www.sciencedirect.com/topics/veterinary-science-and-veterinary-medicine/skeletal-fluorosis
- Izuora K, Twombly JG, Whitford GM, Demertzis J, Pacifici R, Whyte MP. Skeletal fluorosis from brewed tea. J Clin Endocrinol Metab. 2011 Aug;96(8):2318-24.
- Oregon State University. Linus Pauling Institute. Tea. https://lpi.oregonstate.edu/mic/food-beverages/tea#cardiovascular-disease-prevention
Cathy Hester Seckman, RDH, worked in dentistry 32 years, including 12 years as a pediatric hygienist. Officially retired from clinical hygiene, she still fills in occasionally at the same pediatric practice. She is multipublished in dental magazines, works part-time as an indexer, and is the author of three novels, more than a dozen short stories, and an Arcadia Publishing history of her hometown. Her new book, Ohio Day Trips, will be published by AdventureKEEN on March 1.
#1: Maintain Excellent Oral Hygiene
#2: Eat a Nutritious Diet (And Hydrate)
#3: Rinse After Eating Sugar
#4: Visit a Holistic Dentist
About Dr. Thomas
Sources
| Age | Male | Female | Pregnancy | Lactation |
|---|---|---|---|---|
| Birth to 6 months | 0.01 mg | 0.01 mg | ||
| 7–12 months | 0.5 mg | 0.5 mg | ||
| 1–3 years | 0.7 mg | 0.7 mg | ||
| 4–8 years | 1 mg | 1 mg | ||
| 9–13 years | 2 mg | 2 mg | ||
| 14–18 years | 3 mg | 3 mg | 3 mg | 3 mg |
| 19+ years | 4 mg | 3 mg | 3 mg | 3 mg |
Sources of Fluoride
Food
Brewed tea typically contains higher levels of fluoride than most foods, depending on the type of tea and its source, because tea plants take up fluoride from soil [1,3]. Fluoride levels can range from 0.3 to 6.5 mg/L (0.07 to 1.5 mg/cup) in brewed tea made with distilled water [3].
Fluoride concentrations in breast milk are so low that they cannot always be detected; when these levels can be measured, they range from less than 0.002 to 0.01 mg/L, even when mothers live in communities with fluoridated water [3]. Fluoride concentrations in cow’s milk are also very low, ranging from 0.007 to 0.086 mg/L [3]. Fluoride levels in infant formulas in the United States vary, depending on the type of formula and the fluoride content of the water used to prepare the formula [3]. The typical fluoride concentration is less than 0.2 mg/L in milk-based infant formula and 0.2 to 0.3 mg/L in soy-based infant formula (not including contributions from tap water used to prepare the formula).
Only trace amounts of fluoride are naturally present in most foods, and most foods not prepared with fluoridated water provide less than 0.05 mg/100 g [1,6].
A variety of types of foods and their fluoride levels per serving are listed in Table 2.
| Food | Milligrams per Serving |
|---|---|
| Tea, black, brewed, 1 cup | 0.07 to 1.5* |
| Coffee, brewed, 1 cup | 0.22* |
| Bottled water with added fluoride, 1 cup | 0.19 to 0.24* |
| Shrimp, canned, 3 ounces | 0.17 |
| Raisins, ¼ cup | 0.08 |
| Oatmeal, cooked, ½ cup | 0.08* |
| Grapefruit juice, ¾ cup | 0.08 |
| Potatoes, russet, baked, 1 medium | 0.08 |
| Rice, cooked, ½ cup | 0.04* |
| Cottage cheese, ½ cup | 0.04 |
| Pork chop, baked, 3 ounces | 0.03 |
| Yogurt, plain, low-fat, 1 cup | 0.03 |
| Lamb chop, cooked, 3 ounces | 0.03 |
| Tortilla, flour, 1 tortilla, approx. 10” diameter | 0.02 |
| Corn, canned, ½ cup | 0.02 |
| Beef, cooked, 3 ounces | 0.02 |
| Tuna, light, canned in water, 3 ounces | 0.02 |
| Cheese, cheddar, 1½ ounces | 0.01 |
| Bread, white or whole wheat, 1 slice | 0.01 |
| Asparagus, cooked, 4 spears | 0.01 |
| Chicken, cooked, 3 ounces | 0.01 |
| Milk, fat-free or 1%, 1 cup | 0.01 |
| Apple, raw, with skin, 1 medium | 0.01 |
| Avocado, raw, ½ cup sliced | 0.01 |
| Macaroni, plain, cooked, ½ cup | 0.00* |
| Tomato, raw, 1 medium | 0.00 |
| Bananas, 1 medium | 0.00 |
| Egg, cooked, 1 large | 0.00 |
| Carrots, raw, 1 medium | 0.00 |
| Peanut butter, 1 tbsp | 0.00 |
*Amounts of fluoride might vary by levels in the water used to prepare these foods and beverages.
Fluoridated drinking water
Since 1962, the U.S. Public Health Service has recommended the addition of fluoride to drinking (tap) water to reduce the risk and severity of dental caries, one of the most common chronic diseases in children [7]. Many countries around the world now adjust the fluoride concentration of community drinking water supplies to the level recommended for the prevention of dental caries [8].
Although the U.S. Public Health Service recommended fluoride concentrations of 0.7 in warmer climates (where children were expected to drink more water) to 1.2 mg/L in cooler climates to prevent dental caries in 1962, it amended its recommended level in 2015 to 0.7 mg/L to maintain the ability to prevent caries while minimizing the risk of dental fluorosis [7,9]. In 1986, guidelines from the U.S. Environmental Protection Agency (EPA) established a maximum allowable concentration of 4.0 mg/L fluoride in public drinking water systems to prevent adverse effects from fluoride exposure (such as bone disease) and a recommended maximum concentration of 2.0 mg/L to prevent dental fluorosis [3,10]. A review of this regulation is a currently a low priority for the EPA [11].
Fluoridated municipal drinking water—including water that people drink as well as foods and beverages prepared using municipal drinking water—accounts for about 60% of fluoride intakes in the United States [3,7]. In 2016, 62.4% of the U.S. population had access to a fluoridated community water system [12]. The fluoride additives used to fluoridate drinking water in the United States are fluosilicic acid, sodium fluosilicate, and sodium fluoride [13]. The Centers for Disease Control and Prevention publishes fluoride levels in tapwater by county [14]. Because of differences in amounts of fluoride in groundwater, private water sources (including well water) have variable fluoride concentrations [10].
Fluoride is not typically added to bottled drinking waters. However, when fluoride is added, the bottle’s label is not required to list the amount unless the label makes a claim about the product’s fluoride content [15]. By law, the amount of fluoride added to bottled water cannot exceed 1.7 mg/L [3]. In 2019, FDA proposed revising the quality standard for bottled water and thereby limiting the amount of fluoride to no more than 0.7 mg/L, but this rule has not yet been finalized [16].
Dietary supplements
Only a few dietary supplements contain fluoride, usually in the form of sodium fluoride [17]. Most of these products are multivitamin/multimineral supplements, multivitamins plus fluoride, or supplements containing trace minerals only. Some fluoride supplements, usually intended for children, are in the form of drops. The most common amount of fluoride in supplements is 0.25 mg, although a few products contain 0.5 or 1 mg [17].
Dental products
Most toothpaste sold in the United States contains fluoride in the form of sodium fluoride or monofluorophosphate, most commonly at a level of 1,000 to 1,100 mg/L (about 1.3 mg in a quarter teaspoon, a typical amount of toothpaste used for one brushing) [3]. The amount of fluoride ingested from toothpaste depends on the amount used, the person’s swallowing control, and how often the person uses toothpaste. Estimated typical amounts of fluoride ingested daily from toothpaste are 0.1 mg to 0.25 mg for infants and children aged 0 to 5 years, 0.2 to 0.3 mg for children aged 6–12 years, and 0.1 mg for adults [3]. Fluoride in toothpaste, regardless of its form, is well absorbed [1].
Other dental products that provide fluoride include mouth rinses for home use, topical fluoride preparations applied in dentists’ offices or through school-based programs, and dental devices (e.g., orthodontic bracket adhesives, glass-ionomer and some composite resin dental restorative materials, and some dental sealants and cavity liners) [3,18]. Gels used by dentists are typically applied one to four times a year and can lead to ingestions of 1.3 to 31.2 mg fluoride each time; varnishes are least likely to produce a high bolus of fluoride [3].
Medications
Medications can contain fluoride. For example, voriconazole (VFEND or Vfend) is an oral antifungal medication used to treat several infectious conditions, including invasive aspergillosis, candidemia, and candidiasis [19]. Typical doses of voriconazole provide 65 mg/day fluoride. Long-term use (e.g., for 4 months or more) of this medication can lead to high fluoride concentrations in serum [20,21]. The prescribing information for voriconazole advises discontinuation of voriconazole if skeletal fluorosis and periostitis (inflammation of the membrane surrounding and protecting the bones) develop [19].
Fluoride Intakes and Status
Most people in the United States consume adequate amounts of fluoride through foods containing naturally occurring fluoride, fluoridated tap water, and food products made with fluoridated tap water. According to the EPA, typical daily fluoride intakes in the United States from foods and beverages (including fluoridated drinking water) are 1.2 to 1.6 mg for infants and toddlers younger than 4 years, 2.0 to 2.2 mg for children aged 4–11 years, 2.4 mg for those aged 11–14 years, and 2.9 mg for adults [10].
Fluoride and Health
This section focuses on two conditions in which fluoride might play a role: dental caries and bone fractures.
Dental caries
Dental caries occurs when cariogenic bacteria in the mouth ferment foods and produce acids that dissolve tooth mineral [22]. Over time, this tooth decay can cause pain and tooth loss. Without treatment, dental caries can cause infections, impair growth, lead to weight gain, affect school performance, impair quality of life, and possibly result in death [23-26]. Adequate fluoride intakes reduce the risk of dental caries in its initial stages by inhibiting demineralization and the activity of bacteria in dental plaque and by enhancing tooth remineralization [24].
Impact of water fluoridation on dental caries: Water fluoridation protects teeth in two main ways—by preventing the development of caries through ingestion of drinking water during the tooth-forming years and through direct contact of fluoride with teeth throughout life [27,28].
A 2015 Cochrane review included 20 prospective observational studies (most conducted before 1975) [8]. The results showed that water fluoridation reduces the risk of decay and fillings, as well as of premature loss of primary (baby) teeth, by 35% and loss of permanent (adult) teeth by 26% in children receiving fluoridated water in comparison with children receiving unfluoridated water. Fluoridation also increases the number of children with no decay in their baby teeth by 15% and the number of children with no decay in their permanent teeth by 14%. The authors concluded that water fluoridation is effective for reducing dental caries rates in both primary and permanent teeth in children. However, the reviewers were unable to assess the effectiveness of water fluoridation for preventing caries in adults because no evidence met the review’s inclusion criteria (which required studies to include at least two groups, one receiving fluoridated water and one receiving unfluoridated water).
The Cochrane review’s findings were confirmed by a 2018 cross-sectional study on the associations between fluoridated community water and dental caries in the United States [29]. The authors analyzed data on 7,000 children aged 2 to 8 years and 12,604 children and adolescents aged 6 to 17 years who participated in the National Health and Nutrition Examination Study (NHANES) from 1999 to 2004 and 2011 to 2014, respectively. The results showed that living in a county in which 75% or more of the drinking water contained at least 0.7 mg/L fluoride was associated with a 30% reduction in the rate of caries in primary teeth and a 12% reduction in the rate of caries in permanent teeth.
Some evidence shows that the addition of fluoride to drinking water can also prevent dental caries in adults. An observational study included 3,779 individuals in Australia aged 15 and older who participated in the Australian 2004–2006 National Survey of Adult Oral Health [30]. In adults exposed to fluoridated community water supplies for at least 14 years, rates of decayed, missing, or filled teeth were 11–12% lower than in adults whose drinking water during this period had negligible amounts of fluoride. An earlier study in 876 Australian Defence Force members aged 17–56 years found that the average rate of decayed, missing, and filled teeth was 24% lower in those with access to water containing 0.5 to 1 mg/L fluoride for at least half of their lifetime than in those exposed for less than 10% of their lifetime [31].
These findings show that fluoridated drinking water can prevent dental caries in children and adults.
Impact of fluoride dietary supplements on dental caries in children: Some studies have assessed the impact of fluoride supplements on caries development in children. For example, a 2011 Cochrane review of 11 randomized or quasi-randomized studies in a total of 7,196 children (most living in communities lacking access to fluoridated drinking water) found that 0.25–1 mg/day supplemental fluoride for 24–55 months reduced rates of decayed, missing, and filled tooth surfaces by 24% [32]. The authors concluded that fluoride supplements were associated with a lower caries incidence rate in permanent teeth. A 2013 systematic review found an even greater preventive effect of fluoride supplements on the basis of one randomized and four nonrandomized clinical trials in children [25]. The results showed that 0.25–1 mg/day fluoride supplementation reduced caries incidence rates in primary teeth by 48–72% in areas where water fluoridation levels were lower than 0.6 mg/L. In two of these trials that monitored the children for 6–10 years, supplements were associated with a 33–80% reduction in the incidence of caries at ages 7–10 years.
The U.S. Preventive Services Task Force (USPSTF) and the American Dental Association have issued fluoride supplement recommendations for children whose water supply contains little or no fluoride [23]. These recommendations are summarized in Table 3.
| Source | Age Range | Recommendation |
|---|---|---|
| USPSTF [23] | 6 months and older | • Fluoride supplement (dose not specified) for children whose water supply contains little or no fluoride* |
| American Dental Association [33]** | 6 months to 3 years | • Fluoride supplement (0.25 mg/day) for children whose water supply contains less than 0.3 ppm (0.3 mg/L) fluoride |
| 3–6 years | • Fluoride supplement (0.5 mg/day) for children whose water supply contains less than 0.3 ppm (0.3 mg/L) fluoride • Fluoride supplement (0.25 mg/day) for children whose water supply contains 0.3 to 0.6 ppm (0.3 to 0.6 mg/L) fluoride | |
| 6–16 years | • Fluoride supplement (1 mg/day) for children whose water supply contains less than 0.3 ppm (0.3 mg/L) fluoride • Fluoride supplement (0.5 mg/day) for children whose water supply contains 0.3 to 0.6 ppm (0.3 to 0.6 mg/L) fluoride |
*No studies have addressed the dosage or duration of oral fluoride supplementation in this population.
**Recommended doses are based on poor-quality evidence.
Overall, the available evidence suggests that dietary supplements containing fluoride can reduce rates of dental caries in children who lack access to fluoridated drinking water. No studies have assessed the impact of fluoride supplements on caries development in adults.
Fluoride dietary supplements in pregnant women: Like other nutrients, fluoride is transferred from a pregnant woman to her fetus, so a few studies have evaluated the use of fluoride supplements by pregnant women to prevent dental caries in their children. However, the authors of a 2017 Cochrane review found only one randomized controlled trial published in 1997 that met the review’s inclusion criteria [34]. This study assessed caries rates in 798 3-year-old children whose mothers had received 1 mg/day fluoride during the last 6 months of pregnancy [35]. The results showed no significant difference in the proportions of children who had decayed or filled primary tooth surfaces or who had caries. The authors of the Cochrane review concluded that the 1997 study was of very low quality and that no evidence shows that fluoride supplementation in pregnant women prevents dental caries in their offspring.
Bone fractures
Because fluoride helps stimulate the formation of new bone, researchers have hypothesized that fluoride supplements might reduce bone fracture risk. However, research to date has provided only limited evidence supporting this hypothesis [36-38].
The findings of observational studies on the impact of fluoride levels in water on bone mineral density (BMD) and fracture risk have been mixed. A study of 7,129 white women found no significant differences in bone mineral density or risk of hip, vertebral, wrist, or humerus fracture between those exposed and those not exposed to fluoridated water between 1950 and 1994 [37]. In contrast, in a study in 8,266 Chinese residents aged 50 years or older, people with access to water fluoride levels of approximately 1 mg/L had a lower overall risk of fractures, but not of hip fractures, than those with access to water containing negligible fluoride levels [38].
Clinical trials have also had conflicting findings about the efficacy of fluoride dietary supplements to prevent bone fractures. For example, a meta-analysis of 25 randomized controlled trials in a total of 954 participants (four of the studies included people with osteoporosis) showed a significant reduction in vertebral and nonvertebral fracture risk with daily doses of up to 20 mg fluoride (in the form of monofluorophosphate or sodium fluoride), but not with higher doses [39]. A more recent randomized controlled trial found that 2.5, 5, or 10 mg/day fluoride for 1 year in 180 postmenopausal women did not change BMD at any site assessed [40].
Health Risks from Excessive Fluoride
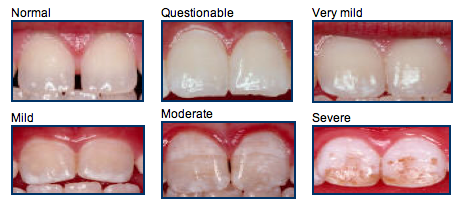
Long-term ingestion of excess fluoride in infancy and childhood, when the teeth are being formed, can lead to dental fluorosis [41]. The characteristics of this chronic condition usually vary from almost imperceptible white lines or flecks to white or brown stains on teeth [2]. Severe dental fluorosis can lead to pitting in tooth enamel. The risk of dental fluorosis increases with fluoride intakes above recommended amounts [42]. Severe enamel fluorosis is very rare, and no evidence indicates that recommended levels of community water fluoridation lead to severe dental fluorosis [3,28].
Analysis of 1999–2004 NHANES data showed that 22.8% of persons aged 6–49 had dental fluorosis, although less than 1% had severe fluorosis and less than 2% had moderate fluorosis [41]. The prevalence rate of dental fluorosis was highest, 41%, in adolescents and lowest, 8.7%, in those aged 40–49. A more recent analysis of NHANES data in 2001–2002 and 2011–2012 found that rates of dental fluorosis (from very mild to severe) increased during this 10-year period, from 29.7% to 61.3% [43].
High doses of fluoride (typically from rare accidents resulting in excessively high levels of fluoridation of water, unintentional ingestion of fluoride products intended for topical use in dentists’ offices, or fluoride supplements inappropriately given to children) can result in nausea, vomiting, abdominal pain, diarrhea, periostitis, and even death in rare cases [3,19,44]. According to one estimate, the acute dose that could cause serious systemic toxicity for fluoride is 5 mg/kg (e.g., 375 mg for someone who weighs 75 kg [165 pounds]) [44]. This dose would be virtually impossible to achieve from water or toothpaste containing standard levels of added fluoride.
Chronic, excess intakes of fluoride are also associated with skeletal fluorosis, although this condition is extremely rare in the United States. Its effects can range from occasional joint pain or stiffness to osteoporosis, muscle wasting, and neurological defects [1,45].
In addition to the potential to damage teeth and bones, some evidence suggests that higher fluoride intakes during early development, including during gestation, might be associated with a lower IQ and other cognitive impairments (e.g., delays in cognitive development) in children [46-49]. However, many experts consider this evidence to be weak and methodologically flawed [50-58].
The FNB has established ULs for fluoride from all sources for healthy individuals (Table 4) based on levels associated with dental and skeletal fluorosis [1].
| Age | Male | Female | Pregnancy | Lactation |
|---|---|---|---|---|
| Birth to 6 months | 0.7 mg | 0.7 mg | ||
| 7–12 months | 0.9 mg | 0.9 mg | ||
| 1–3 years | 1.3 mg | 1.3 mg | ||
| 4–8 years | 2.2 mg | 2.2 mg | ||
| 9–13 years | 10 mg | 10 mg | ||
| 14–18 years | 10 mg | 10 mg | 10 mg | 10 mg |
| 19-51 years | 10 mg | 10 mg | 10 mg | 10 mg |
| 51+ years | 10 mg | 10 mg |
Interactions with Fluoride
Fluoride has no known, clinically relevant interactions with medications [59].
Fluoride and Healthful Diets
The federal government’s 2020–2025 Dietary Guidelines for Americans notes that “Because foods provide an array of nutrients and other components that have benefits for health, nutritional needs should be met primarily through foods. … In some cases, fortified foods and dietary supplements are useful when it is not possible otherwise to meet needs for one or more nutrients (e.g., during specific life stages such as pregnancy).”
For more information about building a healthy dietary pattern, refer to the Dietary Guidelines for Americans and the U.S. Department of Agriculture’s MyPlate.
The Dietary Guidelines for Americans describes a healthy dietary pattern as one that:
- Includes a variety of vegetables; fruits; grains (at least half whole grains); fat-free and low-fat milk, yogurt, and cheese; and oils.
- Includes a variety of protein foods such as lean meats; poultry; eggs; seafood; beans, peas, and lentils; nuts and seeds; and soy products.
- Limits foods and beverages higher in added sugars, saturated fat, and sodium.
- Limits alcoholic beverages.
- Stays within your daily calorie needs.
References
- Institute of Medicine, Food and Nutrition Board. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride. Washington, DC: National Academies Press; 1997.
- Touger-Decker R, Radler DR, Depaola DP. Nutrition and dental medicine. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2014:1016-40.
- National Research Council. Fluoride in Drinking Water: A Scientific Review of EPA’s Standards. The National Academies Press, 2006.
- Pessan JP, Buzalaf MR. Historical and recent biological markers of exposure to fluoride. Monogr Oral Sci 2011;22:52-65. [PubMed abstract]
- Rango T, Vengosh A, Jeuland M, Whitford GM, Tekle-Haimanot R. Biomarkers of chronic fluoride exposure in groundwater in a highly exposed population. Sci Total Environ 2017;596-597:1-11. [PubMed abstract]
- U.S. Department of Agriculture. USDA National Fluoride Database of Selected Beverages and Foods, Release 2. U.S. Department of Agriculture, 2005.
- U. S. Department of Health and Human Services Federal Panel on Community Water Fluoridation. U.S. Public Health Service Recommendation for Fluoride Concentration in Drinking Water for the Prevention of Dental Caries. Public Health Reports 2015;130:318-31. [PubMed abstract]
- Iheozor-Ejiofor Z, Worthington HV, Walsh T, O’Malley L, Clarkson JE, Macey R, et al. Water fluoridation for the prevention of dental caries. Cochrane Database Syst Rev 2015:Cd010856. [PubMed abstract]
- U.S. Department of Health, Education, and Welfare. Public Health Service drinking water standards, revised 1962. Washington, DC: PHS Publication No. 956; 1962.
- U.S. Environmental Protection Agency. Fluoride: Exposure and Relative Source Contribution Analysis. 2010.
- U.S. Environmental Protection Agency. Review of the Fluoride Drinking Water Regulation. 2019.
- Centers for Disease Control and Prevention. Water Fluoridation Data & Statistics. 2016.
- Centers for Disease Control and Prevention. Water Fluoridation Additives. 2014.
- Centers for Disease Control and Prevention. My Water’s Fluoride. 2020.
- U.S. Food and Drug Administration. Small Entity Compliance Guide: Revision of the Nutrition and Supplement Facts Labels. 2020.
- U.S. Food and Drug Administration. FDA Announces Proposed Ruling on Fluoride in Bottled Water. 2019.
- National Institutes of Health. Dietary Supplement Label Database. 2020.
- Cressey P, Gaw S, Love J. Estimated dietary fluoride intake for New Zealanders. J Public Health Dent 2010;70:327-36. [PubMed abstract]
- U.S. Food and Drug Administration. VFEND Prescribing Information 2015.
- Barajas MR, McCullough KB, Merten JA, Dierkhising RA, Bartoo GT, Hashmi SK, et al. Correlation of Pain and Fluoride Concentration in Allogeneic Hematopoietic Stem Cell Transplant Recipients on Voriconazole. Biology of Blood and Marrow Transplantation 2016;22:579-83. [PubMed abstract]
- Tan I, Lomasney L, Stacy GS, Lazarus M, Mar WA. Spectrum of Voriconazole-Induced Periostitis With Review of the Differential Diagnosis. American Journal of Roentgenology 2018;212:157-65. [PubMed abstract]
- Featherstone JD. Dental caries: a dynamic disease process. Australian Dental Journal 2008;53:286-91. [PubMed abstract]
- Moyer VA, Force USPST. Prevention of dental caries in children from birth through age 5 years: US Preventive Services Task Force recommendation statement. Pediatrics 2014;133:1102-11. [PubMed abstract]
- Institute of Medicine. Advancing Oral Health in America. National Academy of Science, 2011.
- Chou R, Cantor A, Zakher B, Mitchell JP, Pappas M. Preventing dental caries in children <5 years: systematic review updating USPSTF recommendation. Pediatrics 2013;132:332-50. [PubMed abstract]
- Kim JK, Baker LA, Davarian S, Crimmins E. Oral health problems and mortality. J Dent Sci 2013;8. [PubMed abstract]
- National institute of Dental and Craniofacial Research. Fluoride & Dental Health. 2020.
- Community Preventive Services Task Force. Dental Caries (Cavities): Community Water Fluoridation. 2013.
- Slade GD, Grider WB, Maas WR, Sanders AE. Water Fluoridation and Dental Caries in U.S. Children and Adolescents. Journal of dental research 2018;97:1122-8. [PubMed abstract]
- Slade GD, Sanders AE, Do L, Roberts-Thomson K, Spencer AJ. Effects of fluoridated drinking water on dental caries in Australian adults. J Dent Res 2013;92:376-82. [PubMed abstract]
- Mahoney G, Slade GD, Kitchener S, Barnett A. Lifetime fluoridation exposure and dental caries experience in a military population. Community Dent Oral Epidemiol 2008;36:485-92. [PubMed abstract]
- Tubert-Jeannin S, Auclair C, Amsallem E, Tramini P, Gerbaud L, Ruffieux C, et al. Fluoride supplements (tablets, drops, lozenges or chewing gums) for preventing dental caries in children. Cochrane Database Syst Rev 2011:Cd007592. [PubMed abstract]
- Rozier RG, Adair S, Graham F, Iafolla T, Kingman A, Kohn W, et al. Evidence-based clinical recommendations on the prescription of dietary fluoride supplements for caries prevention: a report of the American Dental Association Council on Scientific Affairs. J Am Dent Assoc 2010;141:1480-9. [PubMed abstract]
- Takahashi R, Ota E, Hoshi K, Naito T, Toyoshima Y, Yuasa H, et al. Fluoride supplementation (with tablets, drops, lozenges or chewing gum) in pregnant women for preventing dental caries in the primary teeth of their children. Cochrane Database Syst Rev 2017;10:Cd011850. [PubMed abstract]
- Leverett DH, Adair SM, Vaughan BW, Proskin HM, Moss ME. Randomized clinical trial of the effect of prenatal fluoride supplements in preventing dental caries. Caries research 1997;31:174-9. [PubMed abstract]
- Nasman P, Ekstrand J, Granath F, Ekbom A, Fored CM. Estimated drinking water fluoride exposure and risk of hip fracture: a cohort study. J Dent Res 2013;92:1029-34. [PubMed abstract]
- Phipps KR, Orwoll ES, Mason JD, Cauley JA. Community water fluoridation, bone mineral density, and fractures: prospective study of effects in older women. Bmj 2000;321:860-4. [PubMed abstract]
- Li Y, Liang C, Slemenda CW, Ji R, Sun S, Cao J, et al. Effect of long-term exposure to fluoride in drinking water on risks of bone fractures. J Bone Miner Res 2001;16:932-9. [PubMed abstract]
- Vestergaard P, Jorgensen NR, Schwarz P, Mosekilde L. Effects of treatment with fluoride on bone mineral density and fracture risk–a meta-analysis. Osteoporos Int 2008;19:257-68. [PubMed abstract]
- Grey A, Garg S, Dray M, Purvis L, Horne A, Callon K, et al. Low-dose fluoride in postmenopausal women: a randomized controlled trial. J Clin Endocrinol Metab 2013;98:2301-7. [PubMed abstract]
- Beltran-Aguilar ED, Barker L, Dye BA. Prevalence and severity of dental fluorosis in the United States, 1999-2004. NCHS Data Brief 2010:1-8. [PubMed abstract]
- Bhagavatula P, Curtis A, Broffitt B, Weber-Gasparoni K, Warren J, Levy SM. The relationships between fluoride intake levels and fluorosis of late-erupting permanent teeth. J Public Health Dent 2018;78:165-74. [PubMed abstract]
- Wiener RC, Shen C, Findley P, Tan X, Sambamoorthi U. Dental Fluorosis over Time: A comparison of National Health and Nutrition Examination Survey data from 2001-2002 and 2011-2012. J Dent Hyg 2018;92:23-9. [PubMed abstract]
- Whitford GM. Acute toxicity of ingested fluoride. Monogr Oral Sci 2011;22:66-80. [PubMed abstract]
- Gutteridge DH, Stewart GO, Prince RL, Price RI, Retallack RW, Dhaliwal SS, et al. A randomized trial of sodium fluoride (60 mg) +/- estrogen in postmenopausal osteoporotic vertebral fractures: increased vertebral fractures and peripheral bone loss with sodium fluoride; concurrent estrogen prevents peripheral loss, but not vertebral fractures. Osteoporos Int 2002;13:158-70. [PubMed abstract]
- Green R, Lanphear B, Hornung R, Flora D, Martinez-Mier EA, Neufeld R, et al. Association Between Maternal Fluoride Exposure During Pregnancy and IQ Scores in Offspring in Canada. JAMA Pediatr 2019;173:940-8. [PubMed abstract]
- Grandjean P. Developmental fluoride neurotoxicity: an updated review. Environ Health 2019;18:110. [PubMed abstract]
- Office of Health Assessment and Translation, Division of the National Toxicology Program, National Institute of Environmental Health Sciences, National Institutes of Health. Systematic Review of Fluoride Exposure and Neurodevelopmental and Cognitive Health Effects. 2019.
- Valdez Jimenez L, Lopez Guzman OD, Cervantes Flores M, Costilla-Salazar R, Calderon Hernandez J, Alcaraz Contreras Y, et al. In utero exposure to fluoride and cognitive development delay in infants. Neurotoxicology 2017;59:65-70. [PubMed abstract]
- Canadian Agency for Drugs and Technologies in Health. Community Water Fluoridation: A Review of Neurological and Cognitive Effects. 2019.
- Aghaji QN, Nwabuo CC. Association Between Maternal Fluoride Exposure and Child IQ. JAMA Pediatrics 2020;174:210-1. [PubMed abstract]
- Berezow AB. Association Between Maternal Fluoride Exposure and Child IQ. JAMA Pediatr 2019;174:210-7. [PubMed abstract]
- Bledsoe J, Breiger D, McKeever J. Association Between Maternal Fluoride Exposure and Child IQ. JAMA Pediatrics 2020;174:213. [PubMed abstract]
- Farrow S, Joffe AR. Association Between Maternal Fluoride Exposure and Child IQ. JAMA Pediatrics 2020;174:214. [PubMed abstract]
- Gehani CP, Pollick H, Stevenson RA. Association Between Maternal Fluoride Exposure and Child IQ. JAMA Pediatrics 2020;174:215-6. [PubMed abstract]
- Gong CX, James NE. Association Between Maternal Fluoride Exposure and Child IQ. JAMA Pediatrics 2020;174:212-3. [PubMed abstract]
- Ritchie SJ, Morris AJ, McConway K. Association Between Maternal Fluoride Exposure and Child IQ. JAMA Pediatrics 2020;174:213-4. [PubMed abstract]
- Waugh D. Association Between Maternal Fluoride Exposure and Child IQ. JAMA Pediatrics 2020;174:211-2. [PubMed abstract]
- Natural Medicines. Fluoride. 2019.
The Role of Fluoride in Caries Reversal
Dental caries is an infectious disease caused by the complex interaction of cariogenic (caries-causing) bacteria with carbohydrates (i.e., sugars) on the tooth surface over time. Cariogenic bacteria metabolize carbohydrates for energy and produce organic acids as byproducts. The acids lower the pH in the plaque biofilm.47
The hydroxyapatite of tooth enamel is primarily composed of phosphate ions (PO43–) and calcium ions (Ca2+). Under normal conditions, there is a stable equilibrium between the calcium and phosphate ions in saliva and the crystalline hydroxyapatite that comprises 96% of tooth enamel. When the pH drops below a critical level (5.5 for enamel, and 6.2 for dentin), it causes the dissolution of tooth mineral (hydroxyapatite) in a process called demineralization. When the natural buffer capacity of saliva elevates pH, minerals are reincorporated into the tooth through the process of remineralization.47
Caries is generally considered to be a sub-surface phenomenon. With fluoride treatment, a non-cavitated lesion can be remineralized with fluorapatite and have greater resistance to subsequent demineralization than hydroxyapatite. Even at very low concentrations, fluoride is effective as an anticaries agent.49
In the US, there are three commonly used sources of fluoride in oral care products; sodium fluoride (NaF), sodium monofluorophosphate (SMFP) and stannous fluoride (SnF2). All three of these fluoride sources provide the important F– ion, which both inhibits demineralization and promotes remineralization of damaged tooth mineral. In addition, SnF2 is considered to have unique properties, as it provides efficacy against bacterial acids in addition to its fluoridating benefits.Figure 11. A) Demineralization – the caries formation process. Damage occurs in subsurface regions of the enamel, leaving an intact outer layer on the enamel surface. B) Remineralization – the caries reversal process. Caries is reversed through the process of remineralization, in which calcium, phosphate and fluoride are incorporated in the areas damaged due to demineralization processes, resulting in a stronger, fluoridated mineral.
A) Demineralization – the caries formation process. Damage occurs in subsurface regions of the enamel, leaving an intact outer layer on the enamel surface. B) Remineralization – the caries reversal process. Caries is reversed through the process of remineralization, in which calcium, phosphate and fluoride are incorporated in the areas damaged due to demineralization processes, resulting in a stronger, fluoridated mineral.
Sources:
Formerly Bijlage 143 Scentses.isgezond.nl
https://en.wikipedia.org/wiki/Olaflur
https://en.wikipedia.org/wiki/Amine_fluoride
https://my.clevelandclinic.org/health/drugs/20076-stannous-fluoride-dental-rinse
https://pubmed.ncbi.nlm.nih.gov/16878065/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3144112/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2856465/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6923889/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3698581/
https://www.nature.com/articles/d41586-021-02924-6
https://en.wikipedia.org/wiki/Fluoride
https://www.britannica.com/science/fluorine
http://ichemey.blogspot.com/2013/01/ionic-bonding.html
https://www.webelements.com/compounds/sodium/sodium_fluoride.html
https://fr.wikipedia.org/wiki/Monofluorophosphate_de_sodium
https://chem.nlm.nih.gov/chemidplus/rn/7783-47-3
https://pubchem.ncbi.nlm.nih.gov/compound/Olaflur
https://harmonydentaltx.com/7-side-effects-of-consuming-fluoride-you-should-know-about/




